Jean Van Rampelbergh PhD, VP Clinical & Regulatory at Imcyse SA, introduces Imotopes™, the innovative new immunotherapy approach causing scientists to rethink Type 1 diabetes and its treatment
Severe and chronic autoimmune diseases are some of the most difficult and complex immune system-related diseases to treat. Normally, the immune system guards against bacteria and viruses and sends out an army of fighter cells to protect against these infectious diseases. An autoimmune disease occurs when the immune system mistakenly identifies healthy cells as foreign invaders and begins to attack them.
While the exact cause of most autoimmune diseases is still unknown, a small clinical-stage company from the Walloon Region of Belgium, Imcyse SA, has developed a radically new treatment approach. The biotech company is developing a technology platform based on the discovery of modified synthetic peptides – Imotopes™, which specifically block improper autoimmune responses.
Proprietary technology platform for next generation immunotherapies
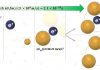
Imcyse’s innovative approach has been validated in numerous successfully completed preclinical and clinical proof-of-principle studies. ImotopesTM are designed and synthetically produced by combining the sequence of a selected HLA class II T cell epitope, which is causally linked to a certain immune-mediated disease, together with a natural amino-acids motif showing oxidoreductase activity. The injection of ImotopesTM generates epitope-specific cytolytic CD4 cells that specifically eliminate antigen-presenting cells as well as other autoantigen-specific lymphocytes of the disease, thereby avoiding autoimmune attacks without affecting other functions of the immune system. Furthermore, induced cytolytic CD4 cells have a memory phenotype, meaning a long-lasting effect can be expected.
Type 1 diabetes and the Imcyse approach
In Type 1 diabetes (T1D), beta-cells in the pancreas are destroyed due to an improper immune response. This damage to the pancreas causes the organ to stop producing insulin, the hormone that controls blood-sugar levels. In most cases, the onset of T1D occurs in children and adolescents, but it can also affect adults.
Imcyse’s lead candidate, IMCY-0098, is a novel insulin-derived ImotopeTM intended to intervene early in the autoimmune response by stopping the destruction of beta-cells. Through this intervention, the pancreas maintains its natural ability to produce insulin. The Imcyse approach is unique and distinctively different to general tolerance induction or overall “immune-suppression.” With this radically new and highly specific technology, the company aims to interfere early enough in the disease progression so that the improper immune response is halted. Through this approach, patients could be cured of the autoimmune disease caused by the mistaken immune response.
Encouraging results from an early clinical study of the T1D ImotopeTM
In a first in-human trial, phase 1b safety study, IMCY-0098 was found to be safe and well-tolerated. The study:
- Demonstrated an excellent safety profile with no safety issues at any dose and no indication of disease exacerbation.
- Detected CD4+ T cells with cytolytic signatures in IMCY-0098 treated patients.
- Identified certain subgroups of patients with signs of improved clinical responses through the use of artificial intelligence data mining analysis.
- Observed correlations between Imotope™-induced immune responses and clinical improvements.
IMCY-0098 Proof of Concept in Type 1 Diabetes – IMPACT Study
In October 2020, a phase 1b/2a clinical trial to evaluate the immune signature of treatment with the ImotopeTM IMCY-0098 and its effect on the preservation of beta-cell function in adult and adolescent patients with recent onset T1D will start. The IMPACT Study, IMCY-0098 Proof of ACtion in Type 1 Diabetes, is a multicentre, randomised, double-blind, placebo-controlled study in patients with T1D within maximum 9 weeks of diagnosis (defined as the day of first insulin injection) at screening and within a maximum of 12 weeks from diagnosis to randomisation. Study sites will be located in Europe with significant participation from hospital trusts throughout the United Kingdom.
Conducted under an adaptive design approach, the study comprises of two steps:
- Step one will enrol 24 participants, age 18-45, who are randomly assigned (1:1:1) to one of three treatment arms and followed for up to 48 weeks.
- Step two will enrol 60 participants, both adults between 18 and 45 years and adolescents aged 12-17 who will be randomly assigned equally to one of three treatment arms based on best dosing findings from step one and followed up to 48 weeks.
For each participant, the study length will comprise a total of approximately 52 weeks from screening to last planned visit.
Step one study objectives:
- Explore and document in adult T1D patients, the immune signature of the treatment with IMCY-0098 at two different doses.
- Determine best dose and regimen according to the immune signature.
Step two study objectives:
- Evaluate whether the immune signature detected in young adults is also present in adolescents using the best dose/regimen selected from step one.
- Evaluate the predictive effect of the detected immune signature on the preservation of beta-cell function measured as stimulated C-peptide after 48 weeks in the whole study population.
- Confirm the safety features of IMCY-0098, both in young adults and adolescents.
- Internationally renowned expert in the field of T1D and coordinator of INNODIA, Prof. Dr. Chantal Mathieu, will act as principal investigator.
Collaboration with the leading T1D network: INNODIA
Imcyse is a proud member of INNODIA, a global network of academic institutions, industrial partners and patient organisations bringing their knowledge and experience together to fight T1D. With collaboration and support from INNODIA in the IMPACT study, Imcyse will gain greater access to T1D patient populations. In addition, this platform for scientific knowledge and technical approaches exchange will aid Imcyse in refining treatment with ImotopesTM as a safe and effective therapy for patients with early T1D.
A broad pipeline
Looking beyond T1D, Imcyse has developed an ImotopeTM for multiple scleroses (MS), based on Myelin oligodendrocyte glycoprotein. The efficacy of the selected ImotopeTM was demonstrated in several preclinical models. A clinical phase I trial in MS patients should start in Q3 2021. The company also initiated a phase 0 study in neuromyelitis optica (NMO). Furthermore, Imcyse entered into an exploratory research agreement with Pfizer for rheumatoid arthritis (RA). Other early stage, preclinical programs include proof-of-concept studies in allergy, graft vs. host rejection, myasthenia gravis as well as in the prevention of immunogenicity in gene therapy using AAV-Vectors. Recently, Imcyse initiated a new program in Celiac disease.
The Imcyse technology platform has the potential to address a wide range of indications in the vast field of immunology and could represent a next-generation, potentially curative approach to severe, chronic autoimmune diseases for which there are no satisfactory therapeutic alternatives.
*Please note: This is a commercial profile
Contributor Profile
Editor's Recommended Articles
-
Must Read >> Why COVID-19 is a concern for people with diabetes