Our research group has developed a novel lens with which to view cancer patients: using computed tomography (CT) images routinely taken during clinical assessment, to precisely quantify body composition (muscle and adipose) in relation to clinical outcomes (Prado et al 2008, Lieffers et al 2009, Lieffers et al 2011, Martin et al 2013). Apart from the quantity of muscle and fat, image-based approaches also reveal additional features such as excess lipid accumulation within muscle tissue. Therefore, disease associated muscle wasting is characterized by myopenia (loss of muscle) as well as myosteatosis (pathological fat accumulation in muscle); features which may co-exist or be present independent of the other.
My other written articles elaborate on the definitions and measures of myopenia and myosteatosis, and I refer the reader to those (here and here). Briefly, tissues can be identified based on their radiological attenuation characteristics measured in Hounsfield Units (HU). The HU value for muscle and fat are known. The presence of excess fat in muscle can be detected owing to the unique radiation attenuation value of adipose tissue compared to lean soft tissue. The radiological density of muscle will decrease as more fat is present in the muscle tissue. Low muscle radiodensity and elevated intermuscular adipose tissue are two related features of myosteatosis measured non-invasively by the use of CT images. The problem of myosteatosis is illustrated by comparing two patients that lie on the extremes for muscle density.
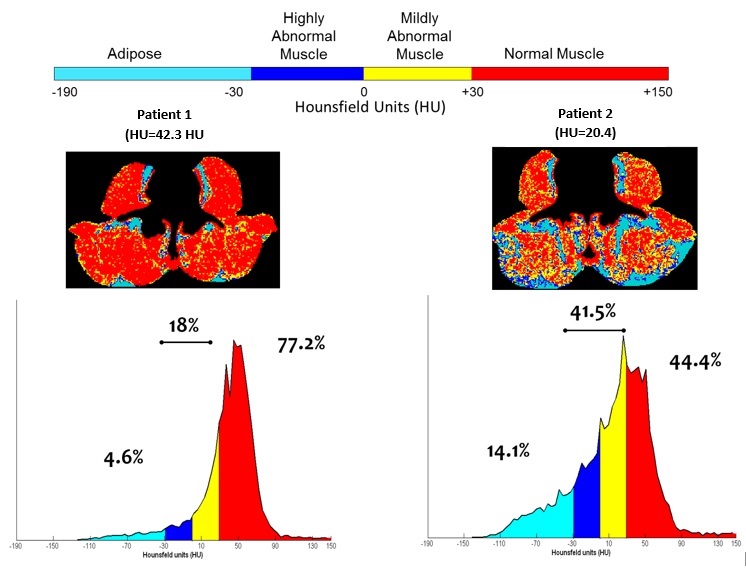
Because CT imaging has emerged as the method of choice for body composition assessment, low radiodensity muscle has only been recently described to exist in the cancer population and predicts markedly shorter survival in every cancer population for which it has been evaluated. The causes and characteristics of low muscle radiodensity and why it confers greater risk is unresolved and continues to be explored by our team in ongoing research. Very little data exists to define the biological, biochemical and physiological features of human muscle that bears the features of myopenia and/or myosteatosis. What is known reveals that poor prognosis and different types of morbidity are predicted by muscle loss and the presence of fat in muscle. While it is not in the usual repertoire of oncologic radiology to report quantifiable dimensions of muscles such as cross-sectional area or attenuation, there may be merit in quantifying skeletal muscle of cancer patients at standard vertebral landmarks, with a view to identify individuals affected by muscle wasting and altered attenuation due to the significant prognostic values these features hold.
References
Aubrey J, Esfandiari N, Baracos V, et al. Measurement of skeletal muscle radiation attenuation and basis of its biological variation. Acta Physiologica. 2014.
Lieffers JR, Sawyer MB Sarcopenia as a prognostic index of nutritional status in concurrent cirrhosis and hepatocellular carcinoma. J Clin Gastroenterol. 2013 Nov-Dec;47(10):861-70. doi: 10.1097/MCG.0b013e318293a825.
Lieffers JR, Bathe OF, Fassbender K, Winget M, Baracos VE.
Sarcopenia is associated with postoperative infection and delayed recovery from colorectal cancer resection surgery. Br J Cancer. 2012 Sep 4;107(6):931-6. doi: 10.1038/bjc.2012.350. Epub 2012 Aug
Martin L, Birdsell L, MacDonald N, et al. Cancer Cachexia in the Age of Obesity: Skeletal Muscle Depletion Is a Powerful Prognostic Factor, Independent of Body Mass Index. Journal of Clinical Oncology. 2013;31(12):1539–1547.
Prado CM, Baracos VE, McCargar LJ, Mourtzakis M, et al. Prevalnce and clinical implications of sarcopenic obesity in patients with solid tumors of the respiratory and gastrointestinal tracts: A population-based study. Lancet Oncol 2008; 9: 629-635.
Dr Vera Mazurak
University of Alberta
Email: vmazurak@ualberta.ca
Website: http://www.albertafamilywellness.org