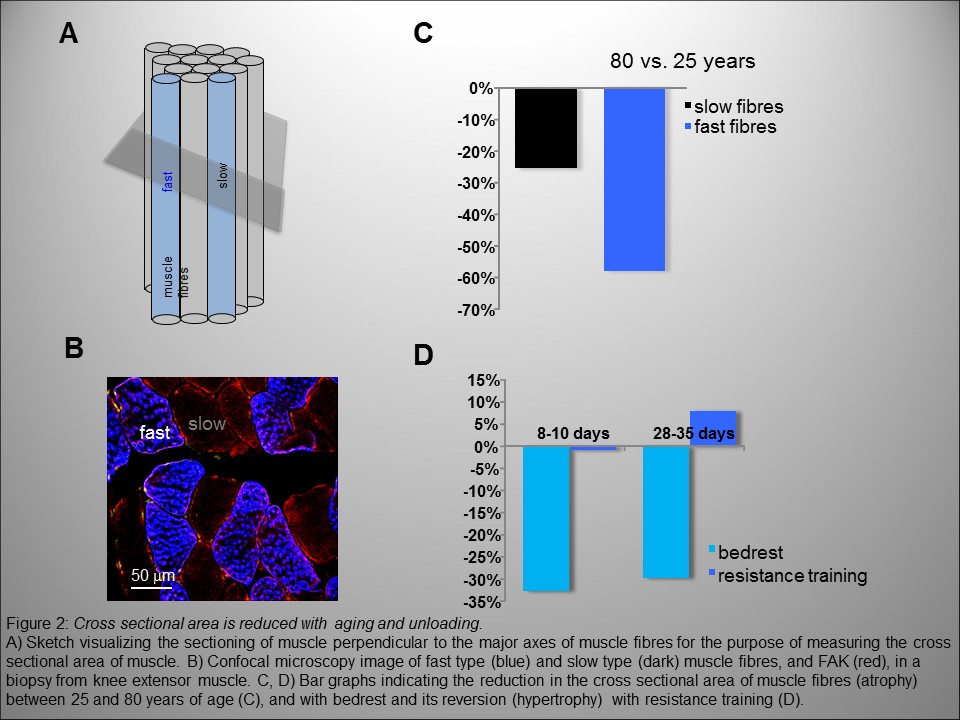
Skeletal muscle functioning is essential for bodily actions and interactions. A number of affections such as injury, disease or the natural course of biological aging, reduce physical performance by provoking a loss in muscle mass (Fig. 1). This phenomenon (called atrophy) has important socio-economic repercussions as a considerable portion of the population is concerned. It is estimated that 500 billion Euros are spent per annum in the European Union, alone, for days of hospitalization days due to muscle weakness.
Mechanical loading is the major physiological regulator of muscle mass beyond development, and even later in life, when the biological processes of aging sets in. This dependence is illustrated by rapid and sustained muscle atrophy in situations that reduce muscle loading, including real of simulated microgravity, or injury of the musculoskeletal system. It is currently thought that 0.5-1% of muscle mass is lost per year beyond the age of 25 years, due to a reduction in the cross section, and possibly length of muscle fibre cells (Fig. 2). This catabolic situation is distinctly, i.e. up to 200-fold, accelerated with muscle unloading after injury of experimental bedrest, where muscle mass is lost at an average rate of 0.5-1% per day for the first two weeks.
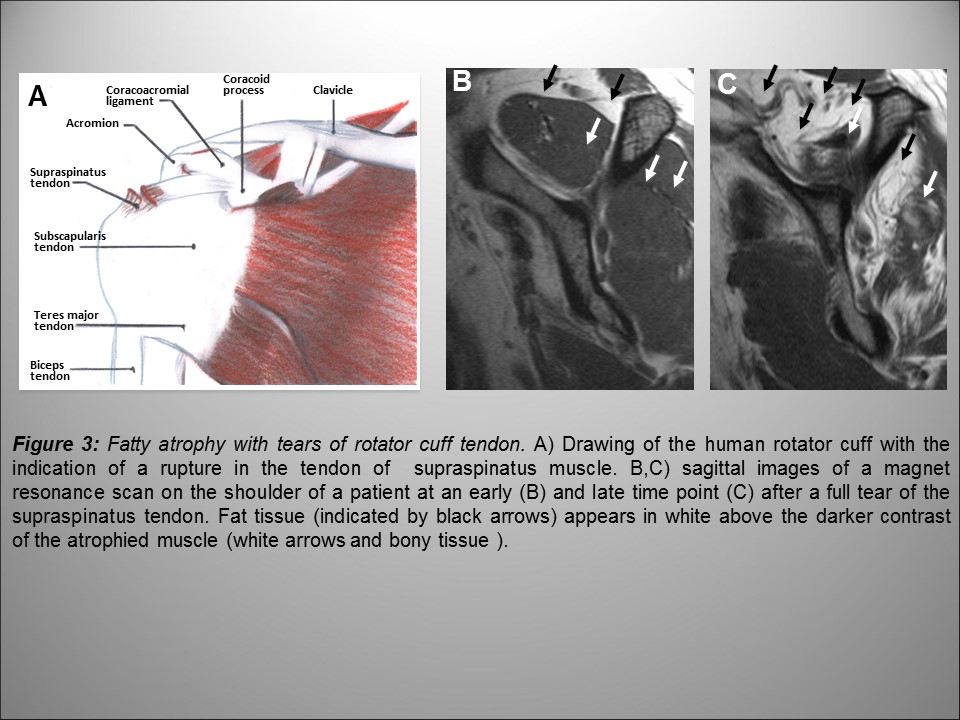
In this regard, muscle atrophy with reduced loading subsequent to tendon rupture with massive muscle overload is particularly devastating. This is because tendon rupture sets a degenerative process in motion where muscle fiber cells are also irreversibly lost and converted into fat (Fig. 3).
Corrective surgery and load bearing contractions during subsequent rehabilitation are the natural remedy to overcome the associated muscle degeneration by activating growth of muscle fibres (hypertrophy). However, in the before mentioned medical situations enhanced muscle loading is not an option in the early, active phase of the pathology or after surgery, due to the incapacity to maintain a sufficient training stimulus because the connection between the muscle-tendon unit is lost or weakened. In other situations such as spaceflight or bedrest, a physiological growth stimulus cannot be provided because the environment does not facilitate to enhance muscle loading. Resolving this insufficiency is a specific priority for the field of Orthopedics, Rehabilitation Science and manned spaceflight where other nutritional and potentially pharmacological options are indicated.
The Laboratory for Muscle Plasticity investigates the molecular processes, which underlies muscle atrophy and its circumvention. This is carried out with the understanding that muscle atrophy involves a shift in the rate and balance between the synthesis (anabolism) and degradation (catabolism) of muscle proteins. Specifically we undertake research into the role of sarcolemmal adhesion complexes (costameres) in orchestrating the balance between biochemical pathways that govern protein synthesis and degradation (Fig. 4). The underlying mechanism involves a double role in the architecture and sensing of mechanical forces. This role is compromised with muscle unloading (Fig. 5).
Our experiments are carried out in collaboration with clinical and pre-clinical partners on models of muscle disuse by unloading. This included human atrophy models, such as bedrest, unilateral limb suspension, space flight, and animal models that permit the modulation of mechano-regulation by (gene) pharmacological and physiotherapeutic means. Bioptic samples are characterized from the levels of genes to contraction using the instrumentalism of functional genomics. Resulting concepts are reintegrated in the context of the orthopedic clinics to moderate, and possible reverse muscle wasting, with clinical affections of the musculoskeletal system.
Laboratory for Muscle Plasticity of the Department of Orthopedics is situated at the University Hospital Balgrist, University of Zurich, Forchstrasse 340, 8008 Zurich, Switzerland
email: mflueck@research.balgrist.ch
Tel: +41 (0) 44 386 3791
Fax: +41 (0) 44 386 3799