Darren K Griffin, Professor of Genetics and Alan R Thornhill Professor of Reproductive Genetics at University of Kent, School of Biosciences lift the lid on why investing in basic embryology research must be a priority
After more than 5 million babies born, in-vitro fertilisation (IVF) is now considered routine. We all know someone who’s had IVF and assume that, because it’s routine, that it’s usually successful. On the contrary, the most likely outcome is failure. The emergence of IVF has led to the development of a number of associated treatments and technologies. In 1990, preimplantation genetic testing arrived with the birth of the Munday twin sisters who were screened for sex as 8-cell embryos because their parents were at risk of transmitting a genetic disease to their sons.
With this breakthrough, came the possibility of screening IVF embryos for chromosomal abnormalities – the most common reason behind IVF failure. Normal human embryos contain 23 chromosome pairs (46 chromosomes total). Those with an abnormal number are termed ‘aneuploid’. The best-known aneuploidy (three copies of chromosome 21) leads to Down syndrome, but other more common consequences include pregnancy loss, congenital abnormality, infertility and, importantly, IVF failure.
Human reproduction is inefficient. Even couples undergoing IVF are at risk of having chromosomally abnormal embryos. Identifying aneuploid embryos, and then implanting only those testing normal (euploid), theoretically minimises these risks to improve IVF outcomes. The procedure is called PGT-A (preimplantation genetic testing for aneuploidy). Those requiring PGT-A are at a greater risk of aneuploid embryos because of advanced maternal age, prior repeated miscarriage or prior repeated implantation failure.
Human reproduction is inefficient. Even couples undergoing IVF are at risk of having chromosomally abnormal embryos.
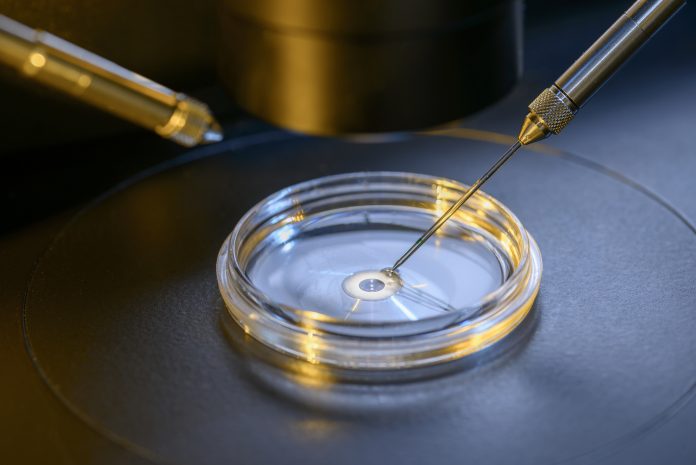
Currently, the test is invasive, requiring the removal of cells from each IVF embryo for genetic testing. This is usually performed on day five or six of development when the embryo has around 100 cells. Removal of five to ten cells at this stage is thought not to harm the future foetus, in part, because the cells removed only represent a small fraction of what will eventually become the placenta.
Early tests for chromosome abnormalities in embryos used a microscopy-based approach with multi-coloured fluorescent probes to identify specific chromosomes in 8-cell stage embryos. However, this method had variable reproducibility, high error rates and not every chromosome was investigated, culminating in many unfavourable randomised controlled trials (RCTs). Considerable in-depth research and development stimulated the introduction of more accurate, sensitive, reproducible tests that simultaneously analysed all chromosomes.
The latest version, next-generation sequencing (NGS), is so sensitive that it detects the presence of chromosomally normal and abnormal cells in the same embryo (so-called mosaicism). Some view mosaicism detection as refined selection, others as a distraction which could lead to the unnecessary discard of potentially normal embryos because low-level mosaics can result in normal live-births.
Since randomised clinical trials (RCTs) over 10 years ago demonstrated that PGT-A could possibly harm (rather than improve) a patient’s IVF chances by discarding potentially normal embryos which test abnormal, there’s been a passionate and polarised debate about whether PGT-A should be performed at all. Authors of the original trials remain fierce opponents, whereas those who benefit directly (and provide benefit by offering PGT-A) remain enthusiastic advocates.
As a result of more recent RCTs, observational studies and large validated national datasets, many IVF practitioners now believe that PGT-A can be beneficial, in the right hands, using the right methods for the right patients.

The anti-PGT-A faction, on the other hand, argues that current evidence is insufficient to justify PGT-A, that any intervention should only be introduced clinically after at least one favourable RCT and even recent studies are biased, limited in size and poorly designed. In response, pro-factions suggest there’s already enough evidence to support PGT-A, it’s unfeasible to conduct large RCTs in some areas of reproductive medicine and many widely used IVF procedures were never subjected to RCTs as their benefits are obvious.
The opposing factions become increasingly entrenched and, for now, the debate continues. Recommendations range from suggesting routine application in all IVF cases to complete discontinuation of PGT-A.
PGT-A can improve but will never be perfect; no test is. What both sides agree on is the need for more research. Chromosomal research on human embryos provides a window of opportunity to study the genetics of the earliest stages of human development, as well as how chromosomes segregate in our sperm and eggs. For example, the mechanisms of how mosaicism arises are poorly understood, despite leading to a range of adverse outcomes, including severe genetic disease in new-borns, stillbirth and miscarriage. This applies not only to IVF embryos but to a significant proportion of naturally conceived foetuses too.
One glaring omission from the scientific literature at present, however, is a comprehensive cell-by-cell comparison of a large cohort of blastocyst embryos to establish overall levels of mosaicism. Taking the research further, we might be able to establish what, if any, approaches can be used to reduce levels of aneuploidy (for instance improved embryo culture (postzygotic) or improved ovarian stimulation or patient management) as there is some circumstantial evidence that some aneuploidy may be induced by the methods and treatments used to perform IVF.
Through in-depth basic research and comparison with model systems (e.g. mouse, pig, cattle), basic questions not only relevant to PGT-A specifically but to IVF in general (and to naturally conceived foetuses) could be addressed more deeply. For instance:
• Are patterns of mosaicism laid down in the embryo or as a result of differential survival of cell lineages?
• If most embryos have some form of (mosaic) aneuploidy, is this normal and what degree of mosaicism can be tolerated?
• Which aneuploid mosaics are least likely to have adverse outcomes?
• Are IVF embryos representative of normally fertilised embryos? This may well not be the case.
• Do chromosome errors perpetuate further errors in human embryos?
In addition to these basic research studies, we need more RCTs, using real-world conditions, some of which stratify patient groups (for example, comparing advanced maternal age versus recurrent implantation failure versus recurrent pregnancy loss etc.).
Future research priorities
Further research is certainly needed to understand how chromosomal abnormalities arise and perhaps how to prevent them. The prospect of non-invasive testing for embryonic aneuploidy using the culture fluid in which the embryo grows to identify cell-free DNA is exciting and recent evidence of this is promising.
The opportunity for understanding aneuploidy and mosaicism in IVF and in the general population that consists mostly of natural conceptions, must not be missed and there has never been a more important time than now to perform basic (human and non-human) research in this area.
We, the authors, would thus like to conclude with this plea: industry, governments, research councils and charities: please work together to generate more funding for basic research into the chromosomes of human embryos.
There are benefits to reap on all sides: For industry, a large proportion of embryos, even from younger women, are aneuploid and will never develop.
We, the authors, would thus like to conclude with this plea: industry, governments, research councils and charities: please work together to generate more funding
Identifying those that will and those that won’t, may lead to a future where every IVF cycle may involve some variant of PGT-A. For the advancement of medical research, a greater understanding of aneuploidy and mosaicism generally will lead to improved patient management and, more importantly, outcomes for many more couples, which with increasingly older families, is one of the most significant genetic problems in reproduction in our time.
References
Darren K Griffin and Çağrı Oğur PGS: a four-letter word?
Biochemist 40(3):26-32.
Griffin DK, Fishel S, Gordon T, Yaron Y, Grifo J, Hourvitz A, Rechitsky S, Elson J, Blazek J, Fiorentino F, Treff N, Munne S, Leong M, Schmutzler A, Vereczkey A, Ghobara T, Nánássy L, Large M, Hamamah S, Anderson R, Gianaroli L, Wells D. Continuing to deliver: the evidence base for pre-implantation genetic screening. BMJ. 2017 Feb 14;356:j752.
Griffin DK, Sheldon S, Introducing PGS into the clinic – “Jacob vs. Giuseppe”. ESHRE Focus on Reproduction November 2017.
Sanders KD, Griffin DK. Chromosomal Preimplantation Genetic
Diagnosis – 25 years and counting. Journal of Fetal Medicine 2017.
Griffin DK, Ogur C. Chromosomal analysis in IVF: just how useful is it? Reproduction. 2018 Jul;156(1):F29-F50. doi: 10.1530/REP-17-0683.
Darren K Griffin
Professor of Genetics
University of Kent, School of Biosciences
Tel: +44 (0)779 302 8711
d.k.griffin@kent.ac.uk
www.kent.ac.uk/biosciences/people/1244/griffin-darren
www.twitter.com/darrenkgriffin
Alan R Thornhill
Professor of Reproductive Genetics
University of Kent, School of Biosciences
Tel: +44 (0)788 049 9617
alan.thornhill@igenomix.com
www.igenomix.co.uk/team
*Please note: this is a commercial profile