Dr Richard Angell and Dr Jane Kinghorn from the Drug Discovery Group (Translational Research Office), at University College London (UCL), explain the value of drug discovery in academia in bridging the “Valley of Death”
The decline in productivity of large pharmaceutical companies over the past two decades and their evolution towards more open and collaborative models has been well documented(1,2). The result of years of downsizing has been the fragmentation of infrastructure required for developing novel small molecules. Highly skilled applied scientists with drug discovery know-how and expertise can now be found in a number of new spin-out therapy companies, Contract Research Organisations (CROs), not-for-profit organisations and also within dedicated Drug Discovery Groups (DDGs) within academia. This new dynamic ecosystem is positioned to actively de-risk early drug discovery through public funding, charities, philanthropy, private-public partnerships and venture capitalist funding. Such collaborative research environments help to overcome common pitfalls in drug discovery where new compounds often fail, known as the “Valley of Death”(5).
Analysis of FDA approved new chemical entities and the role universities have played in their identification and development indicates anything between 24 to 55%(1). Nearly a fifth of drugs recently approved by the EMA originated from academic and publicly-funded drug discovery programmes(3). With large pharma reassessing priorities every two to four years and aligning behind an ever decreasing set of disease areas and indications, large areas of unmet need are now poorly served. Universities are well-positioned to address the gaps. With a diverse set of expertise, access to large sets of data, access to patients, all set in an innovative and creative environment, professionalised DDGs in academia are well placed to advance novel targets, incorporate the latest technologies to de-risk drug discovery projects and progress those projects using robust, industry-standard approaches.
The “Valley of Death” is often described as the gap between a promising early-stage academic discovery and the later-stage de-risked project that will attract the attention of pharmaceutical companies and investors. Bridging this gap can be a significant challenge in an academic environment(5). In a recent report from the ABPI,(4) it was noted that 24 publicly funded drug discovery centres exist in the UK, employing over 500 staff, with the capacity to carry out over 350 screening projects per year. In the U.S., there are approximately 60 such academic translational centres that have received significant funding from the National Center for Advancing Translational Science, National Institutes of Health(5).
Drug discovery at University College London
The Drug Discovery Group (part of the UCL Translational Research Office) is an industry-experienced team of drug discoverers with the task of seeding new drug discovery projects within UCL, focusing on UCL-initiated science. The team collectively has over 40 years of small molecule drug discovery experience across a range of environments from small biotech through to academic groups, research institutes and pharma companies and is positioned within the internationally renowned UCL School of Pharmacy.
Over the past five years, the DDG has established infrastructure to support assay optimisation and cellular and biochemical screening, including the capability to miniaturise assays to 384 and 1,536 well formats and access to screening collections including the AZ Open Innovation library through a strategic collaborative agreement with UCL, brokered by the team. In-house chemistry capabilities include an array of automated purification equipment including Biotage Isolera and Waters prep LC/MS. The team also has extensive experience of out-sourcing research requirements as necessary, including specific hit compound expansion, biophys screening and in vitro and in vivo DMPK studies.
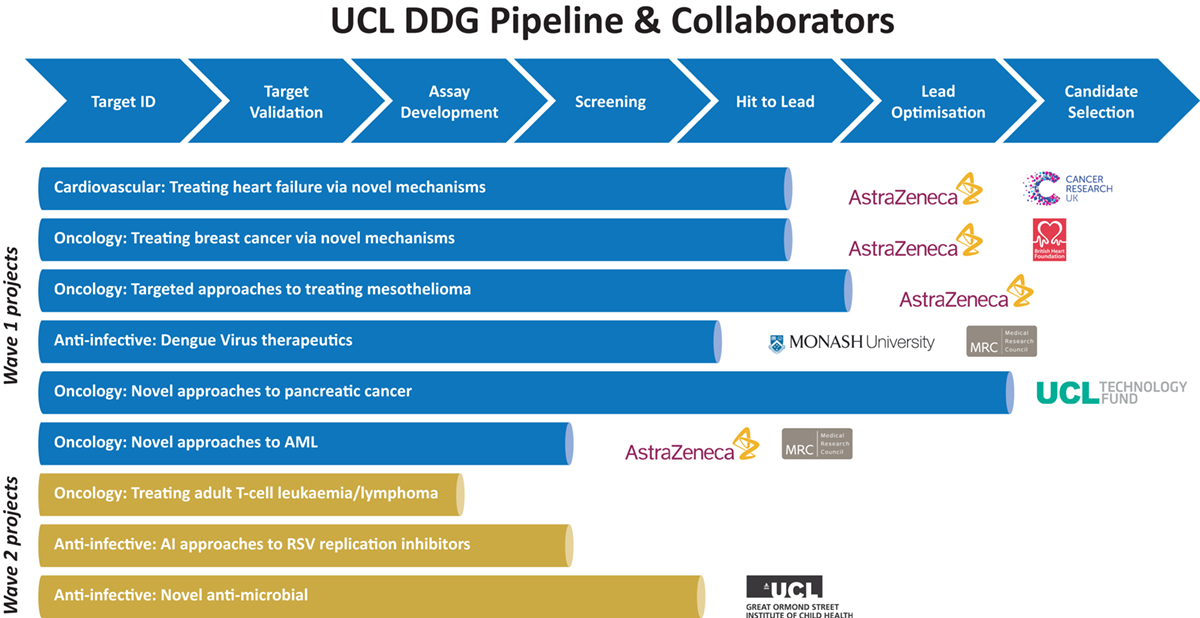
The team are ideally placed to take advantage of UCL’s collaborative ethos between clinician-scientists, patients and academic scientists, translating curiosity-driven academic research into drug discovery projects. This is particularly aided by close ties to our three National Institute for Health Research Biomedical Research Centres (BRCs) providing a unique ecosystem that supports clinical target identification and validation through building a rich repository of patient data and opportunities for experimental and translational medicine. The DDG is currently working with bioinformaticians to enable the intelligent use of data for improvements to target validation and drug design, helping to de-risk the projects the team select to work on.
Current Portfolio
To date, the team has established a portfolio of projects (Wave 1) built upon UCL-based research (Figure). The primary focus has been the cardiovascular, anti-infective and oncology projects, many enabled by Open Innovation collaborations with AstraZeneca. Most of these projects have progressed to validated hit series and are currently seeking on-funding to enable further progression. A second wave of projects are continuing to mature and the team are establishing drug discovery approaches or confirming data associated with early tool compounds.
Funding Models for Academic Drug Discovery
Currently, the team has a mixed funding model with underpinning funds provided by UCLH BRC and Wellcome Trust Infrastructure Strategic Support Fund, supplemented by additional funds from competitively won research applications. These have included awards from UCL Technology Fund, British Heart Foundation, CRUK Drug Discovery Committee, MRC Confidence in Concept and Proximity to Discovery funds.
However, this funding model and, in particular, applying for funding on a project-by-project basis, does not allow the flexibility required to accelerate project progression. A lack of sustainable funding to enable rapid progress in academic drug discovery has been repeatedly recognised in Europe(6), U.S.(7), as well as by the Academy of Medical Sciences in the UK(8) who recently highlighted the need to propagate a culture change in funding for discovery activities to establish more flexible, long-term financial and resource commitments.
Moving forward, the DDG at UCL are actively exploring diversification of their funding model to move towards a sustainable funding stream. The team are exploring flexible portfolio finance opportunities from various sources including VC, public money, charities and philanthropy in return for risk-sharing in the portfolio and identified projects as they mature. If you are interested to participate in exciting, novel early drug discovery, then please get in touch.
References
1 Justin S. Bryans, Catherine A. Kettleborough & Roberto Solari (2019) Are academic drug discovery efforts receiving more recognition with declining industry efficiency?, Expert Opinion on Drug Discovery, 14:7, 605-607.
2 Ajay Gautam and Xiaogang Pan (2016) The changing model of big pharma: impact of key trends, Drug Discovery Today 21:3, 379-384.
3 Helene Lincker, Constantinos Ziogas, Melanie Carr, Nuria Porta & Hans-Georg Eichler (2014) Where do new medicines originate from in the EU?, Nature Reviews Drug Discovery 13; 92–93.
4 The Association of the British Pharmaceutical Industry (2016). The changing UK drug discovery landscape. http://www.abpi.org.uk/publications/the-changing-uk-drug-discovery-landscape
5 Richard J. Barohn, Scott J. Weir, Robert D. Simari,(2019). Progress in Drug Discovery in Academia and Persistent Challenges of “the Valley of Death”, MDMayo Clin Proc. 94(3):391-393 .
6 Bernhard Ellinger & Philip Gribbon (2016) Risk mitigation in academic drug discovery, Journal Expert Opinion on Drug Discovery 11:4, 333-336.
7 Lisa M. Jarvis (2017) Academic drug discovery centers adapt to shifts in funding sources – Universities are pushing their novel molecules into clinical trials Chemical & Engineering News 95:13, 16-18.
8 Academy of Medical Sciences and the Association of the British Pharmaceutical Industry (2016) The UK drug discovery landscape Summary of a joint workshop held on 17 October 2016.
Please note: This is a commercial profile











