Here, Professor Stephen Skinner, Imperial College London, discusses the potential of solid oxide cells to meet our future energy needs with reduced carbon emissions
In June 2019 the UK became the first major international economy to commit to becoming a net zero carbon economy by 2050 [1]. This is a significant change from the previous target of 80% emissions reduction by 2050 and according to the Committee on Climate Change (CCC)[2] the UK was likely to miss the former target. To meet the revised target of net zero emissions by 2050 will require significant technological progress, political will and business engagement. To be clear, net zero emissions means that the UK economy will continue to generate emissions but these will have to be balanced by the removal of greenhouse gases. Whilst the UK was the first G7 economy to legislate for net zero carbon, several other countries have now also committed to net zero carbon, including France and Denmark, whilst the EU has also proposed similar legislation [3].
Whilst these are attractive and essential initiatives it is clear that economies will have to address the key sectors contributing to carbon emissions: Energy Supply (generating electricity), Transport, Business (commercial use of electricity in, for example, manufacturing) and residential (domestic heating). Whilst the energy supply sector has seen significant emissions reductions, the same is not the case in the transport, business and residential sectors, and thus these are a key focus in achieving the net zero economy target. This will require a significant shift in both technology and infrastructure, as highlighted by the CCC [4], including proposals to increase electrification, and develop a hydrogen economy. Developing a clean hydrogen economy using either ‘blue’ or ‘green’ hydrogen will need a diversity of solutions, with one attractive option being the use of solid oxide cells (SOCs) to produce hydrogen, and in the reverse mode, produce combined heat and power (CHP), addressing the domestic heating sector.
Solid Oxide Cell Technology
A solid oxide cell consists of a fuel electrode, an ion conducting electrolyte and an air electrode as the repeat unit, Figure 1, and has the advantage of being scalable, from thin film devices produced through additive manufacturing[5] at the mW power output level through to MW scale stationary devices developed with conventional ceramic processing techniques. In the power generation mode (fuel cell) the devices can use a range of fuels, including methane and hydrogen, with high efficiency, all operating at temperatures in excess of 500 °C. Whilst converting methane would produce CO2 emissions, the high efficiency of these devices means that the CO2 produced per kWh is dramatically reduced, and the SOCs could be integrated immediately into domestic settings. Potential emissions reductions from this immediate implementation are significant, and a step towards zero emissions at the point of use technology. In the longer term this technology is ideally equipped to operate on hydrogen fuel, either in the oxide ion-based device, Figure 1a or in a proton conducting device, Figure 1c. Each of these devices is conceptually identical, with small materials differences between the technologies.
As a hydrogen economy is established, sources for the production of ‘green’ and ‘blue’ hydrogen will be required. Green hydrogen is that produced through renewable technologies combined with electrolysis, whilst blue is hydrogen is generated through reformation of methane. Once again SOC technology offers a solution in the form of high temperature steam electrolysis, that is converting water vapour to a high purity hydrogen stream using a ceramic electrolyser. It is a challenge to undertake high temperature steam electrolysis with low emissions, but as renewable electricity generation has seen considerable growth in recent years with technologies such as solar and wind, combined with storage through battery technologies, steam electrolysis is an increasingly attractive solution. There are two main routes that offer solid oxide electrolysis solutions, and these are based on either an oxide ion conducting (O2-) membrane, Figure 1b, or on proton conducting membranes (H+), Figure 1d.
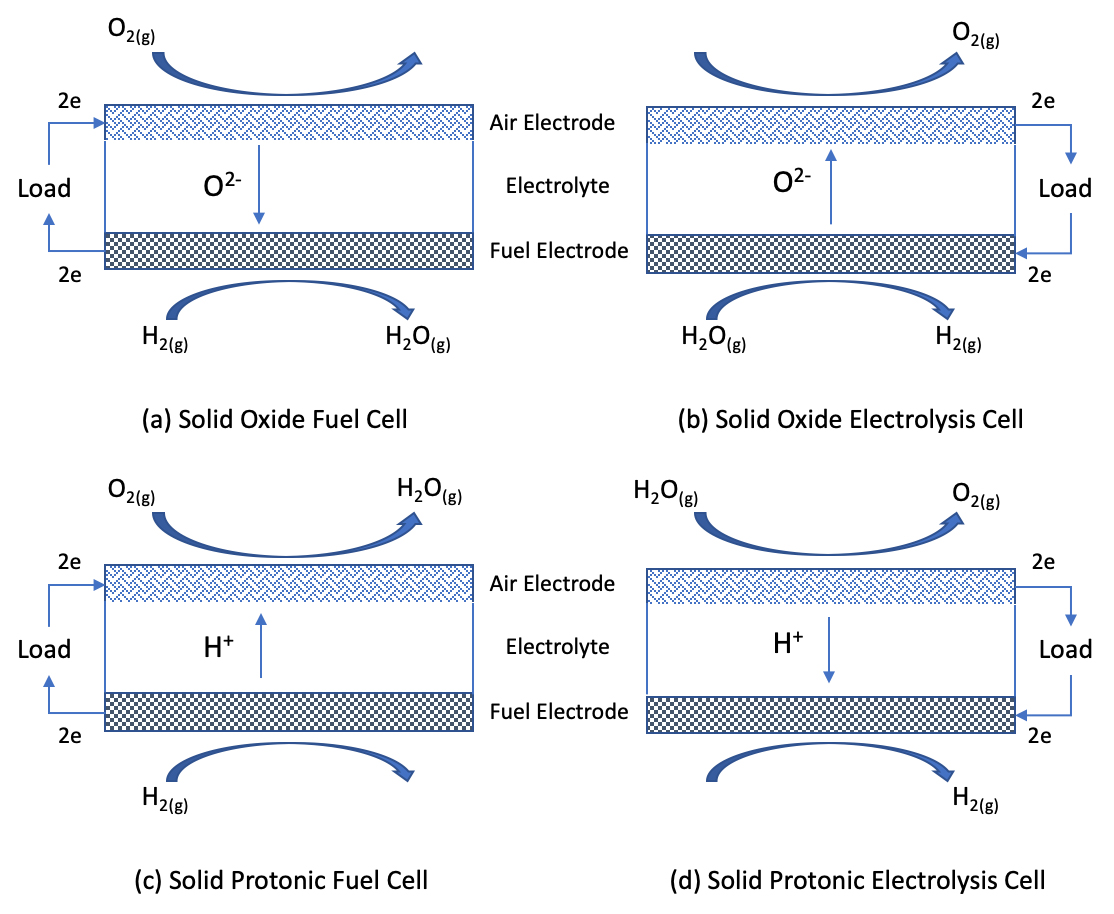
Each of these solid oxide electrolysis cells are in the early stages of development but there are notable successes, with Sunfire offering a product that produces 99.999% H2 with 82% electrical efficiency[7]. Despite this there are considerable challenges that remain to be fully addressed, including the lifetime and durability of devices, degradation phenomena and ultimate performance.
To achieve mass market penetration of solid oxide cells the technology has to achieve some ambitious goals, such as those set by the US Department of Energy of $43 kWh/kg H2 [8] for electrolysis, and to do so requires the development of new high performance materials, combined with in depth understanding of transport processes at, and across, interfaces. Development of effective ceramic electrodes and electrolytes, and optimisation of their nano- and micro-structural features, are essential stages in achieving a net zero carbon economy. Within the field of SOCs there is increasing interest in developing additive manufacturing approaches, allowing innovative new cell designs to be imagined, enabling lower operating temperatures of the cell. These innovations expand the range of application sectors that the technology can service. Supporting fundamental materials science and engineering, combined with technology transfer to systems developers will ensure that rapid progress to meet these ambitious targets can be achieved.
References
[1] https://www.gov.uk/government/news/uk-becomes-first-major-economy-to-pass-net-zero-emissions-law (retrieved 11/8/20)
[2] https://www.instituteforgovernment.org.uk/explainers/net-zero-target (retrieved 11/8/20)
[3] https://ec.europa.eu/commission/presscorner/detail/en/ip_20_335 (retrieved 11/8/20)
[4] https://www.theccc.org.uk/uk-action-on-climate-change/reaching-net-zero-in-the-uk/ (retrieved 11/8/20)
[5] http://www.cell3ditor.eu/ (retrieved 11/8/20)
[6] http://www.h2fcsupergen.com/opportunities-for-hydrogen-fuel-cell-clean-growth-uk/ (retrieved 11/8/20)
[7] https://www.sunfire.de/en/products-and-technology/sunfire-hylink (retrieved 1/9/2020)
[8] https://www.energy.gov/eere/fuelcells/doe-technical-targets-hydrogen-production-electrolysis (retrieved 2/9/2020)
Please note: This is a commercial profile