Here, Professor Stuart Taylor at University College London (UCL), discusses the possibilities of whole body MRI in cancer staging
Following a cancer diagnosis, it is crucial to assess the spread of tumour (if any) to other parts of the body to plan optimal treatment. This spread away from the organ of origin is called metastatic disease. This staging process should ideally be accomplished quickly, with the minimum of risk and inconvenience to patients so their treatment can start as soon as possible. Indeed, delays in cancer diagnosis and treatment are associated with poorer outcomes.
Modern medical imaging platforms including computer tomography (CT), positron emission tomography computer tomography (PET-CT) and magnetic resonance imaging (MRI) are widely used to stage cancer. However, staging pathways can be complicated and time-consuming as often more than one test is required.
This is because imaging tests have different strengths and weakness, so need to be combined. For example, patients diagnosed with lung cancer may need to undergo CT scanning to diagnose the tumour and assess spread to organs such as the liver, PET-CT to stage spread to organs where CT has limited accuracy such as the bones, and MRI to assess the brain, where PET-CT has limitations. Such complex pathways are inconvenient for patients, requiring multiple hospitals visits, exposing them to potentially harmful ionising (X-Ray) radiation, adds to healthcare costs and ultimately delays treatment.
Whole body MRI
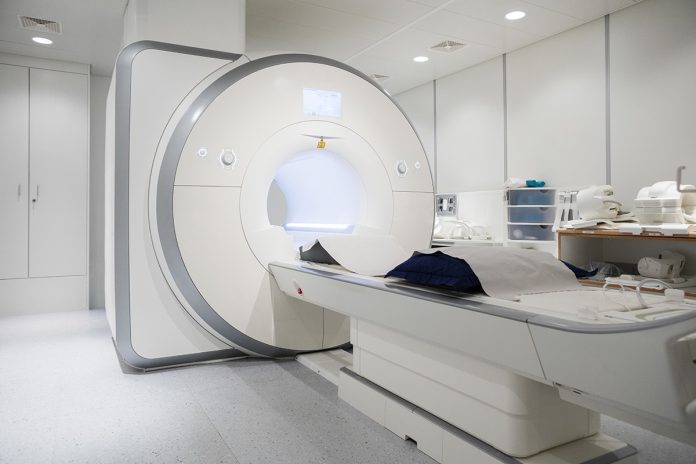
An alternative is whole body MRI (WB-MRI). As its name suggests, the technique scans the whole body (usually from the top of the head to mid-thigh, figure 1) using standard MRI scanners, without exposure to ionising radiation. The principal behind WB-MRI is that it acts as a “one stop shop” staging scan for most patients, speeding up pathways and allowing treatment to begin quickly.
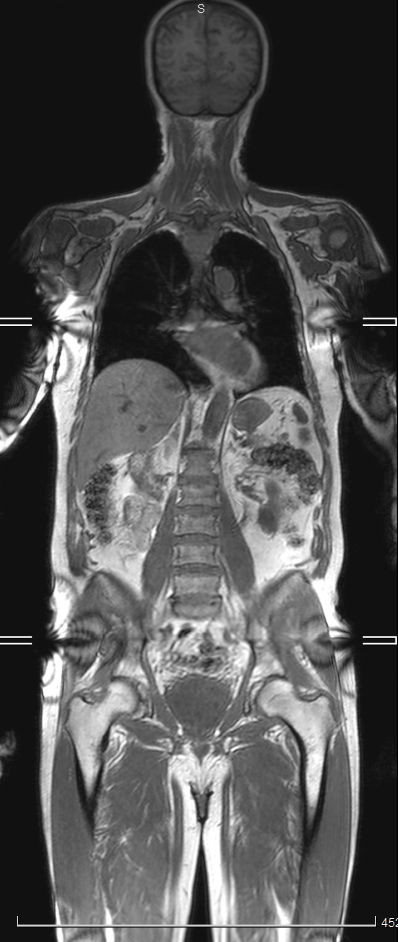
Until recently, however, the evidence supporting WB-MRI was relatively limited. Whilst many studies suggest WB-MRI is as good as or better than standard scans, most include a relatively small number of patients recruited from a single hospital site, with scans analysed by a handful of experienced radiologists.
What was missing were large multisite studies testing WB-MRI as it would be used in large health care systems such as the UK National Health Service (NHS). The National Institute for Health Research (NIHR) specifically commissioned research to address this evidence gap via its Health Technology Assessment programme.
Streamline Trials
The resultant Streamline trials1,2 compared the diagnostic accuracy of initial WB-MRI with standard staging pathways in newly diagnosed non-small cell lung cancer (Streamline L) and colorectal cancer (Streamline C). Both trials ran at 16 English NHS hospitals and 299 (Streamline C) and 187 (Streamline L) patients underwent WB-MRI in addition to their usual staging scans; WB-MRIs were interpreted by 16 and 19 NHS radiologists respectively.
The trials compared complete staging pathways from diagnosis to the first treatment decision made by the multi-disciplinary team meeting, investigating time to completion, scan number needed, costs and impact on treatment plans. As such, the trials considered all the important facets any health care provider needs to know before it invests in new technology such as WB-MRI.
The trials found that for both cancers, pathways using WB-MRI were just as accurate for diagnosing cancer spread (metastatic disease) as standard pathways, resulting in the same treatment decisions, but were quicker (on average by 6 and 5 days for lung and colorectal cancer respectively) and cheaper based on NHS tariff pricing. In the case of colorectal cancer, WB-MRI pathways required significantly fewer tests. Indeed, only 45 (Streamline L) and 21 (Streamline C) additional scans were needed following WB-MRI.
What next for Whole body MRI?
The Streamline trials provide comprehensive and robust evidence supporting WB-MRI as a more efficient approach in staging lung and colon cancer than current standard pathways. This evidence now needs to be reviewed and considered by agencies such the UK national institute for health and excellence (NICE) so that recommendations about the appropriate use of WB-MRI can be made.
Other health care policymakers around the globe should also consider the new data. NICE have already stated that WB-MRI is an acceptable alternative to both CT and PET-CT in staging newly diagnosed multi myeloma. It is not, however, possible to extrapolate results for one cancer type to another. WB-MRI has a potential role in other cancers such as prostate and breast, although robust prospective multisite data is not yet available.
Going forward, health care systems must invest in MRI provision; WB-MRI can be performed on most standard MRI scanners but widespread adoption will require investment in MRI scanner capacity. Radiologists must also be trained in interpretation. Artificial intelligence may play a supportive role in the future, helping to speed up interpretation times while maintaining accuracy.
References
- Taylor SA, Mallett S, Beare S, et al. Diagnostic accuracy of whole-body MRI versus standard imaging pathways for metastatic disease in newly diagnosed colorectal cancer: the prospective Streamline C trial. The lancet Gastroenterology & hepatology 2019; 4(7): 529-37.
- Taylor SA, Mallett S, Ball S, et al. Diagnostic accuracy of whole-body MRI versus standard imaging pathways for metastatic disease in newly diagnosed non-small-cell lung cancer: the prospective Streamline L trial. The Lancet Respiratory medicine 2019; 7(6): 523-32.
This study/project is funded by the National Institute for Health Research (NIHR) Health Technology Assessment programme (10/68/01). The views expressed are those of the author(s) and not necessarily those of the NIHR or the Department of Health and Social Care.
Please note: This is a commercial profile