Stefaan Van Gool, MD, PhD brings to light the important role of Individualised Multimodal Immunotherapy in improving the survival of brain cancer
Despite the intensive research on personalised medicine to treat patients with glioblastoma multiforme (GBM), no real progress has been made in the last decade to improve the prognosis of these patients.
Individualised Multimodal Immunotherapy, having its focus not only on the unique tumour in each patient but also on the tumour microenvironment and the patient’s unique immune system, is a new major step forward to improving long-term overall survival.
Glioblastoma multiforme
Brain cancer remains a rare disease when compared to other cancer types. The term includes more than one hundred different types of tumour entities, categorised in the WHO classification, and regularly updated based on scientific progression. They are distinct from brain metastases. The most frequent malignant primary brain cancer in adults is the GBM. It is an orphan disease, with a yearly incidence of four to six adults per 100,000.
Still, the years of loss of life due to cancer is highest for GBM, compared to other cancer types. GBM can present already at a younger age, and most if not all patients die. The last major and significant step towards standards of care was realised around 2005-2010. Since then, the standard of care has not changed and consists of adjuvant radiochemotherapy and maintenance chemotherapy (Temozolomide) after neurosurgery.

Personalised medicine for glioblastoma multiforme
A clinical risk profile, expressed as the recursive partitioning analysis (RPA one to six), distinct groups of patients depending on grade III/IV malignant glioma; younger/older than 50y patients; below/above 70 Karnofsky performance index; below/above 27 Mini-Mental status; complete/non-complete resection; optimal/suboptimal radiotherapy. Besides, the MGMT promoter methylation status plays an especially important role in chemo/radio-sensitivity and is a dominating prognostic factor.
The diversity of patients became even more obvious based on epigenetic profiling of GBM pointing to at least six different entities. Finally, the expression of membrane molecules and receptors, and the existence of different intracellular pathways opened the possibility for targeted therapies. The latter is the key for personalised medicine approaches. The penetration through the blood-brain barrier within the tumour reduces the spectrum of available drugs. One challenge to prove the efficacy of novel targeted therapies within controlled trials is the diversity of the patient population making the design of an appropriate control population almost impossible. This challenge and the need for rapid testing of new compounds have been partially solved with the development of novel clinical trial designs with adaptive randomisation arms and/or external control arms. However, another not yet solved but more important major challenge is the heterogeneity within the tumour and the rapidly changing unpredictable predominance of different tumour sub-clones over time.
Keeping a fixed treatment protocol, personalised or not, to treat a patient with a permanently changing tumour biology has no rationale. Both challenges explain in part why clinical research has made no progression during the last decennium.
Glioblastoma multiforme within an immune (micro) environment
The particular tumour micro-environment and tumour-host interaction come on top of these challenges. The connection of the immune system within the brain exists, but has special characteristics. The tumour microenvironment is strongly immune-suppressive, and most GBMs are evaluated as “cold”, without immune cell infiltration. Furthermore, there is a predominance of myeloid cells, partially infiltrated, partially being microglia from the brain itself.
A GBM has even a suppressive influence on systemic immune functioning. Treatments like radiotherapy, chemotherapy and steroids also have negative influences on the systemic immune responses. Again, these negative influences are diverse for each patient. This all means that clinicians have to develop new treatment strategies individualised for each patient at the side of the GBM, at the side of the immune system and at the side of the tumour-host interaction. Treatments should allow adaptations over time, following the individual characteristics of each tumour and patient.
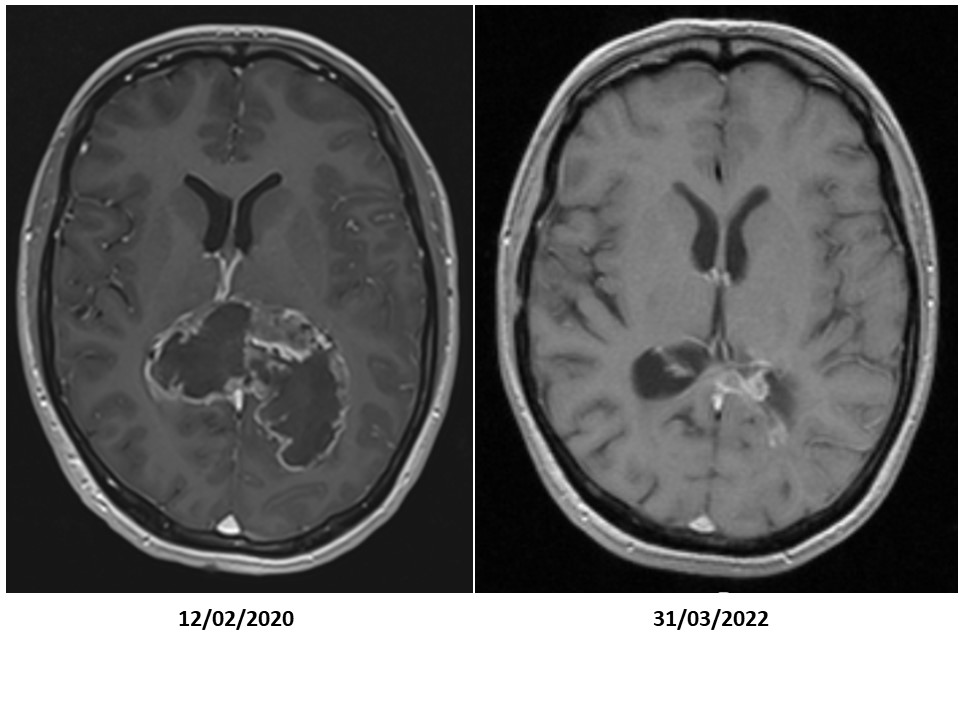
Individualised Multimodal Immunotherapy for glioblastoma multiforme
Neurosurgery and radiochemotherapy are primarily aimed to reduce the tumour volume. Maintenance chemotherapy is aimed to keep control over residual tumour cells. We combined immunogenic cell death therapy (oncolytic virus therapy and modulated electrohyperthermia) during maintenance chemotherapy, and continued with active specific immunotherapy using dendritic cell vaccines and maintenance immunogenic cell death therapy, in combination with modulatory immunotherapy, to improve the overall survival of GBM patients.
To our surprise, a strong synergy became visible between the chemotherapy and the Individualised Multimodal Immunotherapy for the improvement of the overall survival for IDH1 wild-type MGMT promoter-unmethylated patients. Whereas both treatment modalities alone did hardly improve the median overall survival beyond 12 months as obtained with radiotherapy alone, the median overall survival with combination treatment raised to 22 months with a two-year overall survival of 36%. The combination treatment resulted in a median overall survival of 33 months for IDH1 wild-type MGMT promoter-methylated patients with a two-year overall survival of 82%.
To conclude, the role of the Individualised Multimodal Immunotherapy integrated within and after the standard of care to improve long-term overall survival with good quality of life of GBM patients becomes obvious. Immunogenic cell death therapy changes the tumour-host interaction by releasing danger signals within the tumour microenvironment and thereby starting an immunisation process. The dendritic cell vaccines are loaded with antigenic extracellular microvesicles and apoptotic bodies induced by immunogenic cell death therapy, and yielded out of the serum. In this way, the vaccine is loaded with actualised tumour antigens out of residual tumour cells that escaped the radio-chemotherapy and the maintenance chemotherapy. The maintenance immunogenic cell death therapy remains killing potentially appearing new tumour sub-clones thereby including potential novel tumour antigens under the dendritic cell vaccine-induced global anti-GBM immune protection. This treatment strategy is adapted for each patient and each phase of the disease, taking into account dynamic changes in the tumour, in the immune system, and in the tumour-host interaction.
Please note: This is a commercial profile
© 2019. This work is licensed under CC-BY-NC-ND.