With the onset of the COVID-19 andemic, UKAS highlights the importance of maintaining quality control in testing times
The onset of the COVID-19 pandemic forced the government to take quick and decisive action, resulting in the implementation of restrictions on daily lives and business activities. The phrase “Unprecedented times” has entered the lexicon of government, business and the general public alike. Remote working and the curtailing of services has become the new norm for many organisations almost overnight.
During this national emergency, the need for reliable, competent, consistent and verifiable services is as high as ever. In its role as the National Accreditation Body (NAB) for the UK, UKAS has been working closely with both government and conformity assessment bodies (CABs) to ensure that accreditation continues to deliver a globally recognised assessment system which demonstrates that international standards are being met.
Testing times
High-quality testing forms a key part of the government’s five-part plan for managing the COVID-19 pandemic and implementing a gradual easing of restrictions. Accreditation of COVID-19 testing provides a vital level of quality assurance to the process and demonstrates the competence of a laboratory to perform valid test procedures and provide reliable results. Consequently, helping laboratories deliver high quality, reliable tests. As such, UKAS has been closely involved in supporting Public Health England, the NHS and the Department of Health and Social Care in developing accreditation processes that are adjusted to current conditions without compromising standards.
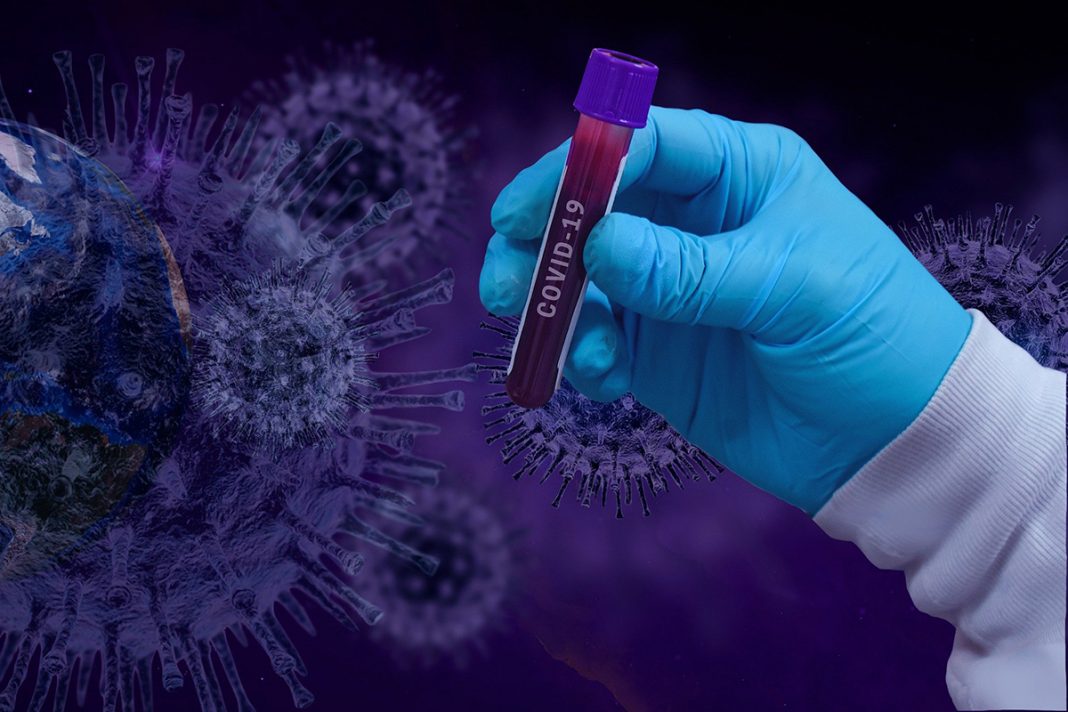
Due to the emergency and the public health demand, it was accepted that this testing for COVID-19 had to commence at pace and before accreditation could be awarded. To help the government’s effort to ensure patients and the public have access to reliable testing, UKAS has implemented a simplified fast track application process for existing accredited laboratories to extend their scope of accreditation. UKAS is receiving applications and enquiries from both public sector and private laboratories that wish to gain accreditation for COVID-19, both for the antigen and antibody testing. The first accreditations for laboratories conducting COVID-19 diagnostic testing were granted at the end of May. Accreditation was awarded against ISO 15189 (the medical laboratories standard) and ISO/IEC 17025 (the general laboratories standard).
UKAS is also providing advice and expertise to the Department of Health and Social Care as a key member of the COVID-19 Testing Capacity Review group. This group of experts is tasked with evaluating the offers received from laboratories to enhance the testing capacity in the coming months.
Assessing the challenge
Accreditation of an organisation provides confidence in the fitness for purpose of an organisation’s services. As such, it is essential that the confidence accreditation provides to government, stakeholders, CABs, their customers and the general public are not diminished at this time.
All accreditation bodies currently find themselves in the situation of needing to provide this confidence but with their traditional business models of on-site assessment severely restricted. Similarly, where CABs are continuing to provide their services, these are often under difficult circumstances of reduced staffing levels, altered working practices and disrupted supply chains. All of which challenge an organisation’s management system to ensure that their services are still fit for purpose.
Providing Confidence – Remotely
To enable it to continue to provide necessary confidence in conformity assessment services, UKAS accelerated the implementation of an existing project on remote assessments, which commenced on 17th March. Remote assessments will continue to be the primary option for organisations seeking to initiate, maintain or extend their scope of accreditation until at least 1st October 2020, and possibly longer, dependent on government policy.
The implementation of remote assessments maintains the vigilance of assessment and quality assurance under the new COVID-19 restrictions. It also ensures any accredited laboratory or organisations involved in supporting the Health and Social Care sector can maintain its status without interruption, where it is safe and appropriate to do so.
Whilst UKAS is accepting applications for extensions to scope and new accreditations in all areas, priority has understandably been given to maintaining or renewing existing accreditations, as well as applications directly related to the fight against COVID-19 such as antibody and antigen testing.
A helping hand
In these difficult times, UKAS is adopting a collaborative approach to assessments as far as possible, without affecting its independence as the NAB for the UK. To help CABs prepare for remote assessments, UKAS has held a series of technical webinars and produced guidance and FAQ documents, all of which are available to view/download on the UKAS website.
Although the format and timeframe of remote assessments will feel different, the structure, processes and fundamental nature of accreditation assessments remains the same.
The confirmation of ongoing technical competence is the ultimate aim of accreditation, and UKAS is pragmatic about how technical activities can be witnessed during remote assessments. Where neither the preferred method of a live audio-visual stream nor narrated video recording is possible, then either a vertical audit of previous work or technical interviews of authorised staff could be undertaken. If none of these routes are available then the fall-back option is to delay the witnessing element of the assessment until COVID-19 restrictions are lifted.
Together, the introduction and refinement of remote assessments for all CABs, the fast-tracking of COVID-19 testing accreditation applications and UKAS’s close work with the relevant government departments and advisory groups have enabled the government to focus its resources elsewhere in the fight against COVID-19, safe in the knowledge that quality and standards are being maintained.
For further media information please contact Phil Russell in the press office:
T: +44 (0)20 7689 5155
E: ukas@clear-group.co.uk
*Please note: This is a commercial profile




![Europe’s housing crisis: A fundamental social right under pressure Run-down appartment building in southeast Europe set before a moody evening sky. High dynamic range photo. Please see my related collections... [url=search/lightbox/7431206][img]http://i161.photobucket.com/albums/t218/dave9296/Lightbox_Vetta.jpg[/img][/url]](https://www.openaccessgovernment.org/wp-content/uploads/2025/04/iStock-108309610-218x150.jpg)





