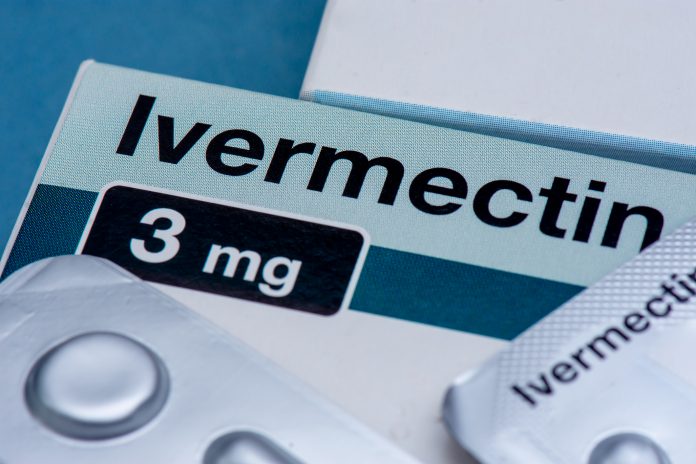
According to a large study, ivermectin does not protect virus patients from the risk of COVID hospitalisation – performing the same as the placebo
In the landmark study, published in The New England Journal of Medicine, researchers find that ivermectin is not useful in the treatment of COVID patients.
Does ivermectin work to stop COVID-19?
No.
The TOGETHER trial, a study team looking at the real-world application of existing drugs to the current pandemic, definitively found that ivermectin does not work. The drug does not lessen the risk of COVID hospitalisation, performing the same as the placebo drug.
The study compared over 1,300 COVID patients in Brazil, who were either given ivermectin or placebo in a double-blind trial.
The authors said: “In this randomized trial, the administration of ivermectin did not result in a lower incidence of medical admission to a hospital or prolonged emergency department observation for Covid-19 among outpatients at high risk for serious illness.”
Ivermectin is normally used to treat parasitic diseases
The World Health Organisation lists ivermectin as a broad spectrum anti-parasitic agent, for use in river blindness, strongyloidiasis and other diseases caused by soil transmitted helminthiasis.
In the US, the drug is also approved to treat external parasites such as headlice and skin conditions such as rosacea.
Ivermectin became a potential miracle drug after “inconsistent” smaller studies
However, ivermectin has been used offhand by medical professionals across the world, after previous studies appeared to confirm benefits.
In 2020, Elsevier published research by Leon Caly, Senior Medical Scientist at the Victorian Infectious Diseases Reference Laboratory (VIDRL) in Australia. This study, proposing that the drug needed further investigation, said: “Ivermectin is FDA-approved for parasitic infections, and therefore has a potential for repurposing.”
However, ivermectin was never approved by the US Food and Drug Administration for COVID. Other, small studies had created enough of a potential benefit to lead the general public into acquiring the drug, while some medical organisations worked to administer it.
Individuals began to self-medicate ivermectin made for livestock
In December, 2021, self-medication became such a risk that the FDA said they had multiple reports of “patients who have required medical attention, including hospitalization, after self-medicating with ivermectin intended for livestock.”
Since livestock ivermectin is created for animals like horses and cows, the dosage can be toxic to the much-smaller frame of a human.
The World Health Organisation (WHO) found that there was very “low-certainty” evidence about what ivermectin can do for COVID patients.
The WHO, echoing the warnings of other health authorities, advised against the use of ivermectin in such patients. After analysing 16 randomised controlled trials, with a total of 2,407 participants, the WHO said only clinical trials should use ivermectin to answer more questions.
It appears that question is now answered – ivermectin does not protect patients from COVID hospitalisation or severe illness.
Despite this scientific output, advocates for the drug continue to push for easier access. An organisation of anonymous nurses in New Jersey, US, tweeted:
Ivermectin can be sold over the counter in TENNESSEE 👏🏻 Cheap effective drugs should always be available to save lives! #Bigwin
— Informed NJ Nurses (@InformedNJNurse) April 25, 2022
Anticipating future rebuke from those that hold onto the myth of ivermectin as a COVID drug, the authors said: “Given the public interest in ivermectin and the support of its use by paramedical groups, we suspect that there will be additional criticism that our administration regimen was inadequate.”